




1 / 5










| Customization: | Available |
|---|---|
| Application: | Pharmaceutical Industry, Industry |
| Capacity: | 400 Gallon |
Featuring Bacteria Prevention & Pressure Stabilization Technology. This sophisticated solution is engineered to meet the stringent demands of pharmaceutical and laboratory settings, ensuring pure and contaminant-free water production.
Mainly used for the storage of raw water for pharmaceutical water treatment and medicine liquid for dosing systems.


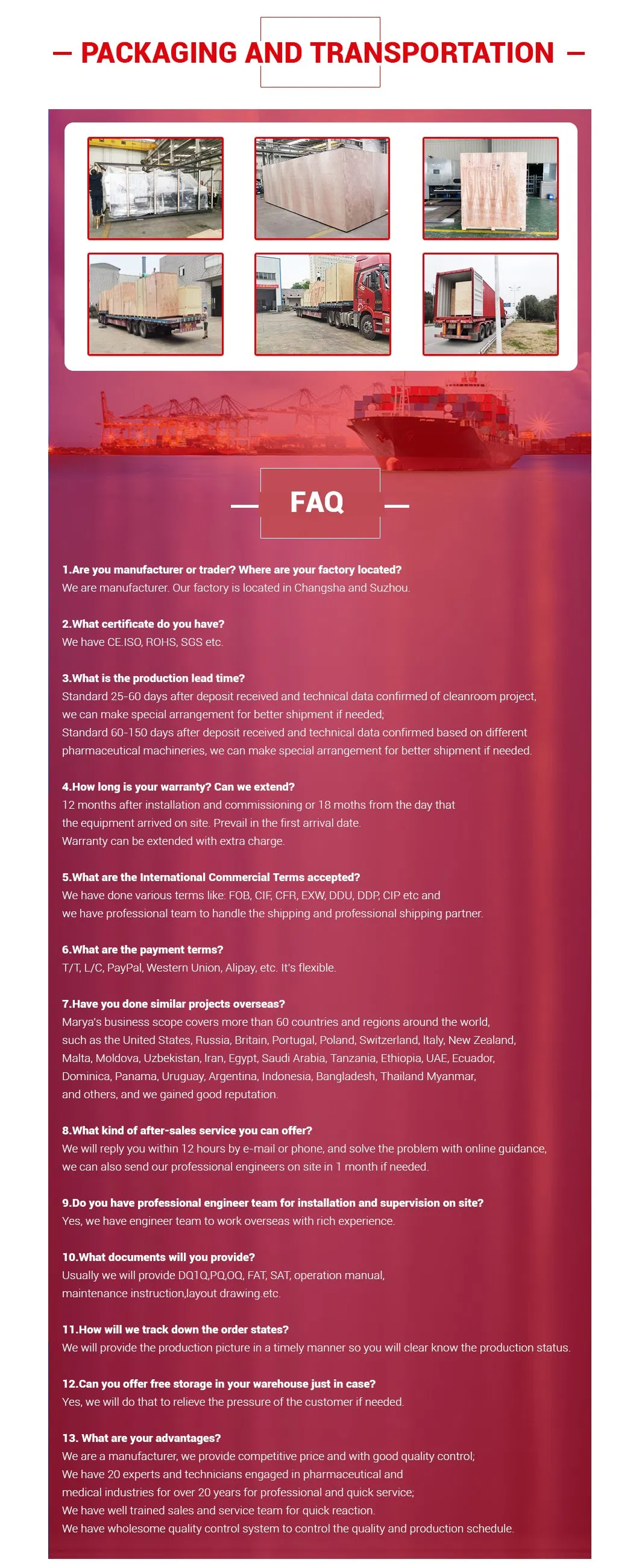
Designed for storage and transport of liquids in pharmaceutical, food, and chemical industries.
High-efficiency membrane technology equipment meeting GMP requirements.


Designed per cGMP, ISPE guidelines, and FDA specifications.
Thermal distillation solution for generating pharmaceutical-grade Water for Injection (WFI).


Generates pharmaceutical-grade pure steam (PS) for high-temperature sterilization.








 Sanva Medical
Sanva Medical